Preweaning mortality can claim up to 20% of piglets born, and there is evidence that the rate is increasing as the industry continues to successfully nudge litter size higher.
February 15, 2013

Preweaning mortality can claim up to 20% of piglets born, and there is evidence that the rate is increasing as the industry continues to successfully nudge litter size higher. A key factor contributing to preweaning mortality is whether piglets nurse successfully after birth.
Most piglets are born with enough energy reserves to survive for about a day, so it is essential that they nurse shortly after birth to acquire the additional nutrients needed to survive.
Fortunately, most piglets are born hungry and begin to search for the sow’s udder almost immediately. The sow does her part, too. Normally, the hormones associated with pregnancy
stimulate the udder to produce colostrum before the birthing process begins. Usually, the interaction between piglet and sow works well and requires nothing more than letting nature take its course.
However, for a variety of reasons, some piglets fail to nurse, which increases their risk of mortality dramatically. Some sows fail to lactate or to make colostrum, which puts the whole litter at risk.
Scientists at the Agricultural Research Service’s U.S. Meat Animal Research Center (USMARC) in Clay Center, NE, have been working on strategies to reduce these failures. They have developed a simple, cost-effective method that could be used to determine whether colostrum was successfully obtained by piglets. This method is called the “immunoglobulin immunocrit” or “immunocrit” for short.
Immunoglobulin Assay
Piglets are born with very low levels of immunoglobulins, which are proteins that defend the piglet from diseases. Colostrum differs from the milk produced later in lactation in that it has very high concentrations of these proteins. Immunoglobulin proteins pass quickly into the blood of newborn piglets after nursing. The ability of the piglet to absorb these proteins only lasts about 24 hours, which means that measurements of immunoglobulins in blood can be made on Day 1 after birth.
The immunocrit assay measures the amount of these proteins in the blood of piglets, and that value can be used to determine if a piglet is getting enough colostrum. By monitoring the immunocrit in all piglets in a litter, it is possible to detect failure of colostrum production by the sow while most of the piglets are still alive. If problems are identified, piglets can be saved by crossfostering or other means.
In their work, scientists have established that immunocrit measurements below a threshold are associated with increased piglet mortality, depending on the size of the piglet.
The assay is relatively simple, requiring a small amount of blood (1 ml. or less), a 40% solution of ammonium sulfate, a hematocrit microcentrifuge tube, a tabletop hematocrit centrifuge (about $1,000) and a ruler.
It turns out that immunoglobulins precipitate in 20% ammonium sulfate solution and most other proteins do not. With equal parts of ammonium sulfate solution and blood, samples are spun in the centrifuge and the length of the precipitate and the solution are measured. The ratio of the two provides a quantitative measure of the immunoglobulins in the sample.
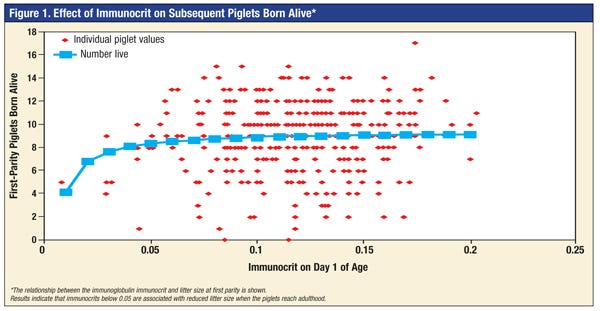
Because pigs are born with essentially no serum immunoglobulins, any immunoglobulins they have originate from colostrum. In other words, this simple assay can tell you if a piglet got colostrum. Ratios below 0.05 are considered “low” and are associated with poor piglet survival. Results indicate that about 4% of piglets are below this threshold (Figure 1). With appropriate training and equipment, the assay could easily be performed on the farm.
Clearly, low-birth-weight pigs are at greater risk for not getting colostrum, but surprisingly, many do just fine. In one collaboration where we related immunocrit measures to visual assessment of piglets as to whether their bellies were full, we learned that visual assessment missed a lot of pigs that did not get colostrum. This is not too surprising when you consider that piglets are typically born with a belly full of amniotic fluid.
The immunocrit assay provides an objective measure of the piglet/sow colostrum interaction and, therefore, can be used to monitor management strategies and improve procedures for optimizing colostrum intake through split-suckling, for example.
Colostrum Affects Future Fertility
On its own, using the immunocrit to reduce preweaning mortality by improving colostrum intake would be useful enough, but there is another dimension to this assay. Recent work by scientists at Auburn and Rutgers Universities indicates that colostrum does more than just provide energy and immunoglobulins to newborn piglets. If piglets with low ratios do survive, they are less likely to be fertile as adults.
The Auburn and Rutgers work has shown that colostrum contains factors that are needed for the normal development of the uterus and cervix in gilts, the testes in boars and other components of their reproductive systems. The reproductive tract tissues of male and female piglets that do not receive colostrum for two days after birth, but still manage to survive, will be altered.
One factor in colostrum that supports development of the female reproductive tract is relaxin, a hormone that was first identified as a factor that helps prepare the adult reproductive tract for birth. However, researchers also know that other unidentified factors are present in colostrum, since treatment with relaxin alone does not fully mimic
the effects of colostrum on tract development.
The process through which the factors in colostrum that affect reproductive tract development are passed from mother to offspring is called lactocrine. The Auburn/Rutgers team has shown that colostrum consumption and, therefore, “lactocrine signaling” is required to support normal patterns of uterine development in newborn gilts.
The effects of lactocrine signaling on reproductive tract development suggest that a lack of colostrum might impair the reproductive capacity of pigs during their adult life. Because the immunocrit can be used to detect piglets that do not receive sufficient colostrum, this new assay was employed to determine the relationship between immunocrit on Day 1 after birth and the fertility in the same gilts as adults.
Results indicated that piglets with low immunocrit values on Day 1 after birth that survived to adulthood had smaller litters. Therefore, the immunocrit assay could be used as a screening tool for the very early removal of animals that are poor candidates for the breeding herd. In gilts, very low immunocrit values on Day 1 after birth would be a good predictor of reduced reproductive capacity as adults due to the lack of colostrum and sub-optimal lactocrine signaling.
Genetic Markers
Further use of the immunocrit will help researchers unravel the factors that contribute to poor piglet nursing ability and failure of sows to initiate colostrum production. Scientists at USMARC have collected immunocrit data on thousands of piglets, and they are using those data to find regions of the pig genome that are associated with failure of piglets to nurse
successfully.
In addition, researchers are using litter average immunocrits to discover genomic regions associated with sow colostrum production. These genetic markers will be useful in selecting for piglets and sows that are more capable of engaging successfully in this essential interaction.
Further research by Auburn University and Rutgers University will be undertaken to clarify the components of colostrum that are essential for the proper development of the reproductive tracts, but also other organ systems that require colostrum for normal development. Collectively, this research will improve the acquisition of colostrum by piglets, resulting in reduced preweaning mortality and improved reproductive and other components of the biology of the adult pig.
About the Authors - Jeffrey L. Vallet is supervisory research physiologist at the U.S. Meat Animal Research Center, Clay Center, NE; Frank F. Bartol is alumni professor and associate dean for research and graduate studies, College of Veterinary Medicine, Auburn University, Auburn, AL; and Carol A. Bagnell is professor and chair of the School of Environmental and Biological Sciences at Rutgers University, New Brunswick, NJ.
You May Also Like


