Why Can’t Some Pigs Walk?
It’s frustrating for producers when they find a pig “down” (Figure 1) or with central nervous system (CNS) signs (Figure 2) whereby pigs cannot walk – their ability to move from place to place being compromised.
April 1, 2013

It’s frustrating for producers when they find a pig “down” (Figure 1) or with central nervous system (CNS) signs (Figure 2) whereby pigs cannot walk – their ability to move from place to place being compromised. Sometimes, the first clue that something is wrong is suddenly finding a pig dead.
Figure 1. "Downer" with Posterior Paresis

Figure 2. " CNS" with Tremors

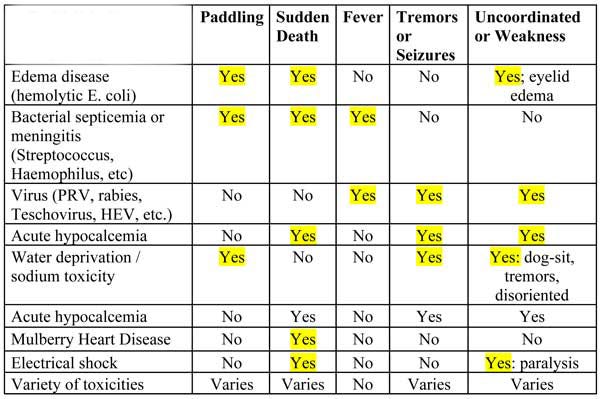
Signs of lameness or locomotory disease can occur at any age from birth to market. Accurately diagnosing the cause is challenging because several body systems can be involved. A short list of common diseases is found in Table 1.
Table 1. Common Diseases of CNS or Sudden Death
The body systems that can contribute to locomotion dysfunction (inability to move with normal gait) include:
1. The nervous system (brain and/or spinal cord and/or nerves);
2. Muscle system;
3. Skeletal system;
4. Joints and tendons; and
5. Hoof and claw.
Often, multiple body systems may be involved in the same pig. Commonly, one can find combinations of maladies in the same group or barn. Accurate assessment of clinical signs can be helpful to differentiate which of the body systems are affected. Moreover, accurate communication of the signs observed is very important for establishing cause.
For example, the term “CNS signs” to Bob, the nursery manager, means laying on the side paddling or “streppy.” But to Jane, the finishing manager, CNS signs mean “downer” pigs. The use of photographs and video are increasingly available to effectively communicate to other production team members what is seen in the barn.
Sometimes the cause can be determined fairly accurately by clinical examination of pigs, but it usually takes laboratory testing to confirm a particular cause and to add confidence in the diagnosis. Certainly not all diseases that impair locomotion are infectious. Any outbreaks of clinical disease that “look different” probably are different and they should be fully investigated via careful examination, necropsy and submission of samples to a laboratory.
Body Systems
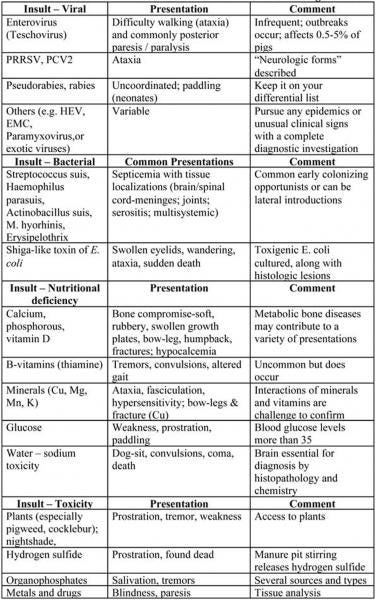
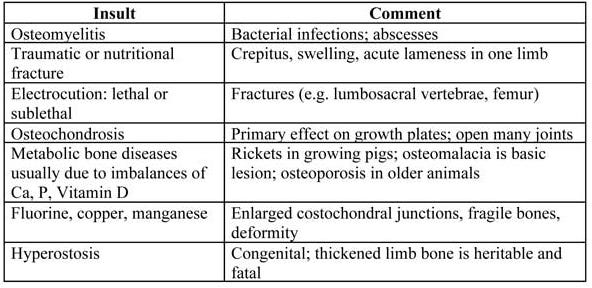
Brain: Diseases that affect the brain usually cause loss of awareness. Affected pigs do not recognize or react to human presence. For example, pigs that lay on their side, perhaps in a coma, with paddling or odd eye movements. With some diseases, pigs may be uncoordinated or have tremors with disease originating from brain lesions. Table 2 contains some diseases affecting brains.
Table 2. Most Common Insults and Clinical Presentations Affecting the Brain
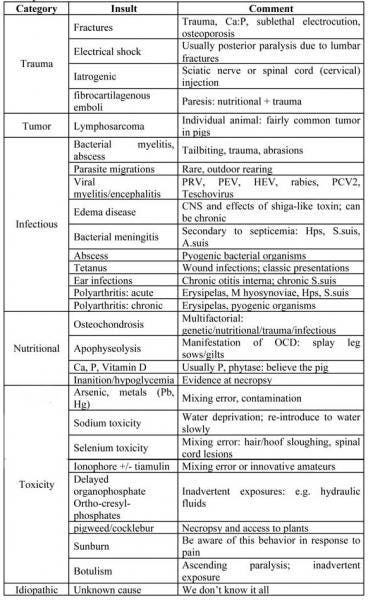
Spinal Cord: Pigs with insults to spinal cord will have central awareness (their brain is unaffected), but cannot walk properly. They may be uncoordinated or may be unable to use their hind and/or front legs properly. In some cases, they simply “dog sit.” Diseases affecting the spinal cord are listed below in Table 3.
Table 3. Conditions that may be Associated with Posterior Paresis or Paralysis
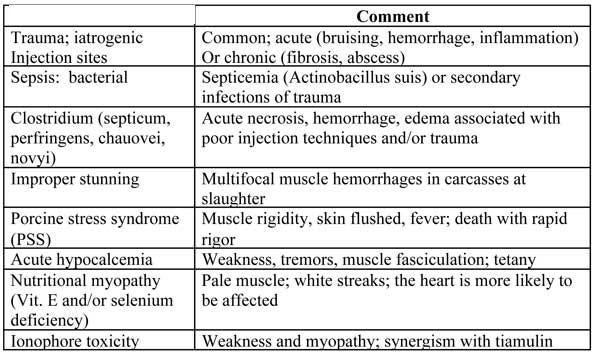
Muscle: While not common, muscle injuries and diseases do occur. Individual muscles may be swollen and overlying skin reddened with bacterial infections such as Clostridium. With generalized diseases (e.g. ionophore toxicity), muscles may be weak, hence, pigs cannot stand and walk. Muscle diseases are found in Table 4.
Table 4. Muscle Insults
Joints: Joint injuries and diseases are very common and can affect single or multiple joints, depending on cause. Pigs are heavily muscled and active; athletic injuries are probably common but difficult to confirm. Individual pigs may have lameness as result of stretched ligaments, tendons, bruises or microfractures similar to human athletes. With outbreaks of lameness, more pigs will have a lameness or joint stiffness. Onset and prevalence may be abrupt or slowly increase. Many joint and bone insults occur over the lifetime of a pig; those effects are probably cumulative.
Joints may be swollen, and if acute bacterial infection is present, may be warm to the touch. When joint disease is suspected, it is important to determine which joints are affected (i.e. stifle, hock, carpal, elbow). Acute joint insults are very painful. Pigs in pain may be savaged by penmates. Unattended, lame pigs cannot compete; they may fall behind and become a welfare issue. When outbreaks occur, a laboratory submission is usually warranted. Table 5 lists some of the common diseases associated with joints.
Table 5. Joints: Common Insults and Presentations
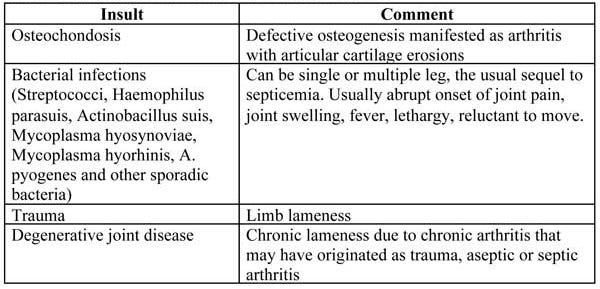
Skeletal system/bone: Metabolic bone diseases (rickets, osteomalacia, osteoporosis) have received increased attention over the last several years. Because of the variation in clinical signs, gross lesions, and physiologic mechanisms with different ages, diagnosis can be problematic. The most reliable indicator for rickets is confirming the presence of soft, rubbery bones (ribs that bend or break easily with very little “snap”) and increased rate of fractures in older pigs. In some age groups, pigs with acute hypocalcemia may have tremors, tetany or simply be found dead. Often signs are progressive from sudden deaths to increased occurrences of weakness, lameness or fractures.
Table 6. Bone: Common Insults and Presentations
Feet and claws: Many of the inciting causes of feet and claw lesions are related to trauma. Inadequate micronutrients, such as biotin, are uncommonly implicated in hoof lesions.
Diagnostic Investigation
Clinical disease in populations is rarely due to a single insult. Therefore, quantifying subtle qualitative changes can be very helpful to sort out causes from effects. The first step is to establish a clinical case definition, which is essentially the primary and early clinical signs that are consistent. Since populations often have more than one syndrome occurring simultaneously, it is useful to create clinical case definition “buckets.” Record the number affected with each clinical sign or bucket per unit (pen, barn). Quantifying the numbers affected will help prioritize diagnostic and intervention strategies.
Like what you're reading? Subscribe to the National Hog Farmer Weekly Preview newsletter and get the latest news delivered right to your inbox every Monday!
The diagnostic process is essentially a matter of detecting and defining deviations from normal by physiologic and anatomic examination, then assigning cause. Necropsy can help sort out some causes of locomotory disease. Some diseases create very characteristic clinical signs, but most do not. Some diseases have characteristic gross or microscopic lesions (e.g. infectious arthritis, suppurative meningitis), but others do not (e.g. tetanus, organophosphates, metabolic hypocalcemia).
When thorough clinical examination or field necropsy is not sufficient for confident diagnosis, laboratory testing is usually warranted. The best time to thoughtfully collect relevant samples would be the present (right now). Considering all the expenses of a laboratory submission – the value of animals sacrificed, potential loss of group productivity, shipping, professional fees, laboratory fees and time – every effort should be made to collect a complete sample set. This cannot be driven by some standard operating procedure (SOP). It requires that a knowledgeable person think and then execute – taking the time to do it right the first time.
What should be collected for laboratory submission? With locomotory diseases, it is helpful to examine both acutely and chronically affected animals. Acutely affected, non-medicated animals are used to confirm primary infectious causes whereby levels of the insulting agent are still detectable and disease is, perhaps, yet uncomplicated. Some lesions are very hard to detect in samples from acutely affected animals, yet are very well developed or even diagnostically evident in chronically affected animals. There may be more than one insult contributing to the problem and it may be useful to identify the progression of insults for more holistic interventions. A fairly complete list of samples to collect is found below. We don’t want the excuse for “no diagnosis” to be that the right stuff was not submitted.
Feed: it costs virtually nothing to collect, label and store a 2-lb. feed sample.
Pool samples from feeders and bins·
Store in cool, dry cabinet or submit
Water: 1 pint sample from pig level (pool) and 1 pint sample from building source
Freeze or submit
Serum and whole blood from affected pigs and asymptomatic cohorts
Serum can be useful for clinical pathology, nutrient analysis, agent and antibody detection, etc.
Whole blood, just in case we have red cell, white cell, toxin concerns
Brain: Fresh and formalin-fixed brain sample
Try not to captive bolt animals with suspected brain problems
Remove with cleaver, hatchet, saw or by splitting the head with a knife in pigs less than 25 lb.
Split brain longitudinally, putting half in formalin and half on ice
Brainstem and cerebellum samples are very important
Collect as much of the spinal cord as you can with the brain
A swab of the brain stem can be collected but it alone is not sufficient
Sending a fresh, chilled intact head is better than not submitting brain at all
Spinal cord: fresh and formalin-fixed,1-in. segments of all parts of the cord
Remove spinal cord with Barnes dehorner, cleaver or saw
Alternatively, knife-cut intervertebral discs to free up individual or clusters of vertebrae
Sending intact vertebrae with the spinal cord inside is better than no spinal cord at all
Muscle and heart: fresh (tennis ball size) and fixed (thumb-drive size)
Affected muscle, multiple sites; semimembranosus, longissimus, psoas when in doubt
Assures you have looked outside and (hopefully) inside the heart
Bone: routinely break a few ribs; if in doubt, collect bones and tissues
Collect serum (affected and cohorts labeled)
Collect second rib, costochondral growth plates, enlarged joints, liver, full set of tissue
Reminder: Collect some feed
Joints: submit either intact joint or joint fluid and synovium (fresh and fixed)
Sample joints aseptically, then open them (vs. contaminating them first)
Remove overlying skin
Use syringe and needle to aseptically collect synovial fluid
Open joint, examine, collect synovium (and articular surface if OCD is suspected)
Submitting intact, chilled joints; be sure you actually send the joints from affected legs!
Hoof and claw: intact foot is fine
Describe and photograph
Collect anything you see that is unusual: varmints, rodents, flies, weeds, bags, tags, nostrums.
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)

